  
Polonium, for example, is dangerous due to its toxicity and radioactivity. Some are found in drugs, cleaners, and pesticides. Metalloids are extremely important in electronics as semiconductors, although they are also used in optical fibers, alloys, glass, and enamels.As far as appearances go, metalloids range from dull to shiny.The electronegativity and ionization energy values of metalloids. Oxidation numbers of elements in this group range from +3 to -2. For this reason, these Frankenstein-type elements are also called semimetals or semiconductors. Metalloids may either gain or lose electrons during chemical reactions.One exception is silicon, which is brittle. Semimetals are typically malleable and ductile. 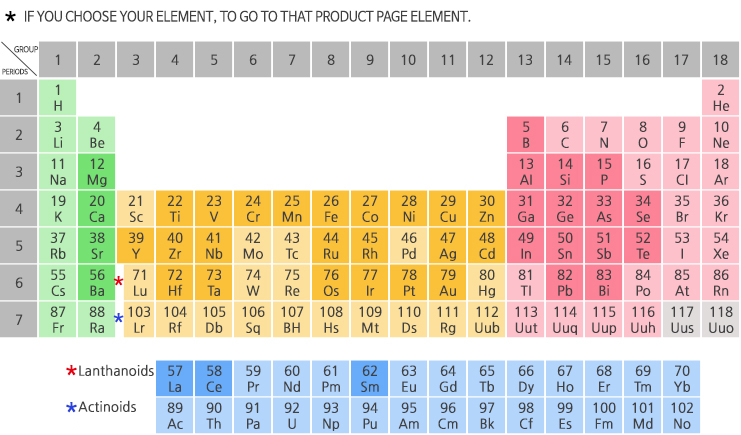
Semimetals/metalloids have high lattice dielectric constants and high diamagnetic susceptibilities.Some metalloids, such as silicon and germanium, can act as electrical conductors under the right conditions, thus they are called semiconductors. 
Metalloids are all solid at room temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
